α2-3,6,8,9 Neuraminidase A
α2-3,6,8,9 Neuraminidase A is a broad specificity sialidase, which cleaves linear and branched non-reducing terminal sialic acid residues from glycoproteins, glycopeptides, and oligosaccharides. It can be used for glycan analysis and characterization and intact glycoprotein remodeling, in vitro and in vivo.
- Recombinant enzyme with no detectable endoglycosidase or other exoglycosidases contaminating activities
- Acts on both Neu5Ac and Neu5Gc
- Double digest with other exoglycosidases and endoglycosidases
- Tolerant of moderate levels (0.5-1.0%) of detergents
- ≥95% purity, as determined by SDS-PAGE and intact ESI-MS
- Optimal activity and stability for up to 24 months

-
Product Information

Neuraminidase is the common name for Acetyl-neuraminyl hydrolase (Sialidase). α2-3,6,8,9 Neuraminidase A catalyzes the hydrolysis of all linear and branched non-reducing terminal sialic acid residues from glycoproteins and oligosaccharides. The enzyme releases α2-3 and α2-6 linkages at a slightly higher rate than α2-8 and α2-9 linkages.
Specificity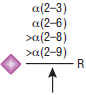
Detailed Specificity
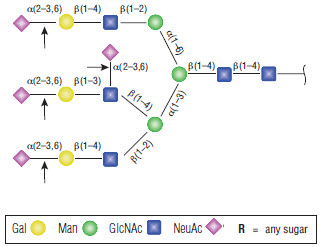
α2-3,6,8,9 Neuraminidase A will cleave branched sialic acid residues that are linked to an internal residue. This oligosaccharide from fetuin is an example of a side-branch sialic acid residue that can efficiently be cleaved (1).
Product Source
Cloned from Arthrobacter ureafaciens and expressed in E. coli.- This product is related to the following categories:
- Exoglycosidases Products,
- Proteome Analysis Products
- This product can be used in the following applications:
- Expression Systems,
- Glycan Sequencing,
- Proteomics,
- Recombinant Glycoprotein Expression, Glycoprotein Analysis
-
Protocols, Manuals & Usage
-
Tools & Resources
-
FAQs & Troubleshooting
-
Citations & Technical Literature
-
Quality, Safety & Legal
Other Products You May Be Interested In
Ineligible item added to cart
Based on your Freezer Program type, you are trying to add a product to your cart that is either not allowed or not allowed with the existing contents of your cart. Please review and update your order accordingly If you have any questions, please contact Customer Service at freezers@neb.com or 1-800-632-5227 x 8.